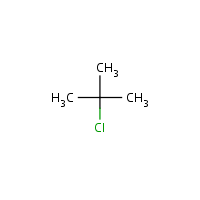
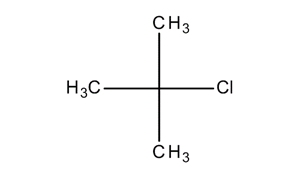
Explain the relative reactives of butyl chloride, sec-butyl chloride, and tert-butyl chloride with ethanolic AgNO3 in term of the structure of the transition state | Homework.Study.com

Is the tert-Butyl Chloride Solvolysis the Most Misunderstood Reaction in Organic Chemistry? Evidence Against Nucleophilic Solvent Participation in the tert-Butyl Chloride Transition State and for Increased Hydrogen Bond Donation to the 1-Adamantyl
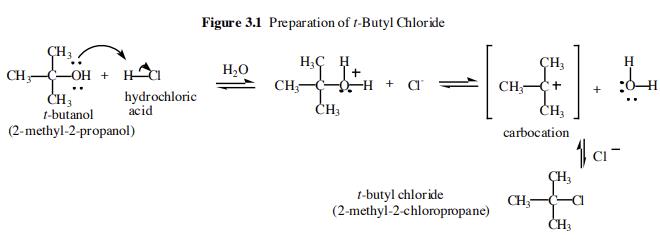
A solvent-catalyzed four-molecular two-path solvolysis mechanism of t-butyl chloride or bromide in water or alcohol derived by density functional theory calculation and confirmed by high-resolution electrospray ionization-mass spectrometry | SpringerLink

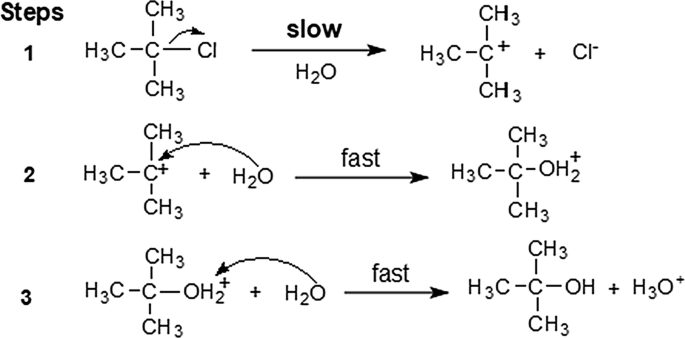
Some tert-butyl ether is formed in the solvolysis of tert butyl chloride. Where does this product come from (i.e. what is the substrate and what is the nucleophile?) Do you think this